 
The electrons are free to travel great distances (hence the conductivity of metals) and serve as a glue to hold all of the positively charged metal nuclei together. Finally, in metals the outermost electrons are donated or "pooled" in the band structure that exists in metals. The bonds formed through electrostatic attraction are not as strong as those formed from covalent sharing of electrons. The ionic atoms are attracted to one another through electrostatic attraction and the crystal lattices that are formed. In ionic materials such as sodium chloride, the electrons are donated from one (the electropositive) atom to the other (the electronegative) atom in order for the atoms to achieve a filled shell structure. In covalent bonds such as those in methane and oxygen, the valence electrons are shared between the atoms involved in the bond and they (the electrons) spend most of their time in the region between the nuclei involved in the bond this makes for a strong bond. What you learned in your mineralogy class was correct bond strength decrease in the following order covalent > ionic > metallic. 
However, when covalent bonds form network covalent structures, atoms combine to form a singular macromolecule that is much stronger than singular covalent bonds. You can see this in the fact that the boiling points of ionic salts are much higher than that of a covalent substance like water. 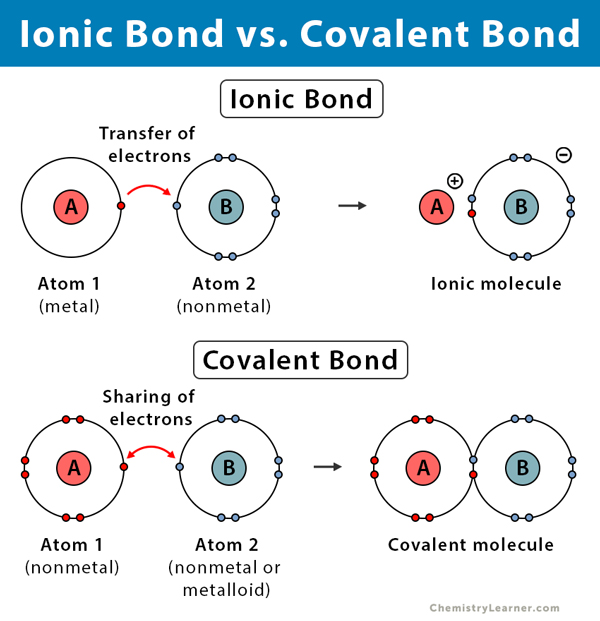
So to answer your question, substances with standard covalent bonds seem to be weaker than those with ionic bonds because the ionic bonds tend to form a lattice structure, that makes them much stronger. The network structure combines to make the substance stronger than normal covalent bonded substances. Some other structures you might want to look into are Graphite and Graphene, which are both allotropes of carbon (allotropes are, simply put, different molecular arrangements of an element). This molecular network is also the reason that diamond and quartz form a crystalline structures, just like you'd see in ionic substances such as NaCl. These structures form a lattice-like structure, much the same as ionic compounds. Quartz and diamond are stronger substances because their molecules form network covalent structures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
